Dynamics of Toxoplasmosis Disease in Cats population with vaccination
Keywords:
Toxoplasmosis, Vaccination, Cats, Sensitivity indexAbstract
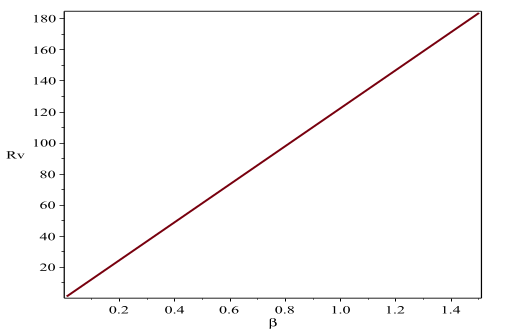
We extend the deterministic model for the dynamics of toxoplasmosis proposed by Arenas et al. in 2010, by separating vaccinated and recovered classes. The model exhibits two equilibrium points, the disease-free and endemic steady states. These points are both locally and globally stable asymptotically when the threshold parameter Rv is less than and greater than unity, respectively. The sensitivity analysis of the model parameters reveals that the vaccination parameter $\pi$ is more sensitive to changes than any other parameter. Indeed, as expected the numerical simulations reveal that the higher the vaccination rate of susceptible individuals the smaller the value of the threshold Rv (i.e., increase in $\pi$ results in the decrease in Rv , leading to the eradication of toxoplasmosis in cats population.
Published
How to Cite
Issue
Section
How to Cite
Similar Articles
- E. A. Nwaibeh, M. K. M. Ali, M. O. Adewole, The dynamics of hybrid-immune and immunodeficient susceptible individuals and the three stages of COVID-19 vaccination , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 3, August 2024
- Ogechi Regina Amanso, Jeconia Okelo Abonyo, Phineas Roy Kiogora, Obiora Cornelius Collins, A novel mathematical model for transmission dynamics of HPV and cervical cancer progression with cancer-reliant awareness , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 2, May 2026
- L. O. Afolagboye, Z. O. Arije, A. O. Talabi, O. O. Owoyemi, Effect of Pre-Test Drying Temperature on the Properties of Lateritic Soils. , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 1, February 2023
- J. Andrawus, J. Y. Musa, S. Babuba, A. Yusuf, S. Qureshi, U. T. Mustapha, A. Oghenefejiro, I. S. Mamba, Modeling the dynamics of pertussis to assess the influence of timely awareness with optimal control analysis , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- Danat Nanle Tanko, Farah Aini Abdullah, Majid K. M Ali, Matthew O. Adewole, James Andrawus, Onchocerciasis control via Caputo-Fabrizio fractional dynamics: a focus on early treatment and vector management strategies , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 1, February 2026
- Isah Charles Saidu, Musa Yusuf, Florence Chukwuemeka Nemariyi, Ayenopwa Comfort George, Indexing techniques and structured queries for relational databases management systems , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 4, November 2024
- Abiola T. Owolabi, Kayode Ayinde, Taiwo J. Adejumo, Wakeel A. Kasali, Emmanuel T. Adewuyi, Comparative Analysis of the Implication of Periods Before and During Vaccination of COVID-19 Infection in Some Regional Leading African Countries , Journal of the Nigerian Society of Physical Sciences: Volume 4, Issue 2, May 2022
- Bolarinwa Bolaji, Abdullahi Ibrahim, Favour Ani, Benjamin Omede, Godwin Acheneje, A model for the control of transmission dynamics of human monkeypox disease in Sub-Saharan Africa , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 2, May 2024
- Akeem Olarewaju Yunus, Morufu Oyedunsi Olayiwola, The analysis of a novel COVID-19 model with the fractional-order incorporating the impact of the vaccination campaign in Nigeria via the Laplace-Adomian Decomposition Method , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 2, May 2024
- Olumide S. Adesina, Adedayo F. Adedotuun, Kayode S. Adekeye, Ogbu F. Imaga, Adeleke J. Adeyiga, Toluwalase J. Akingbade, On logistic regression versus support vectors machine using vaccination dataset , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 1, February 2024
You may also start an advanced similarity search for this article.






