Investigations of the Elastic Moduli of Er2O3 NPs Doped TeO2 – B2O3 – SiO2 Glasses using Theoretical Models
Keywords:
Erbium oxide, Tellurite Glass, Elastic Moduli, Poisson Ratio, Theoretical ModelsAbstract
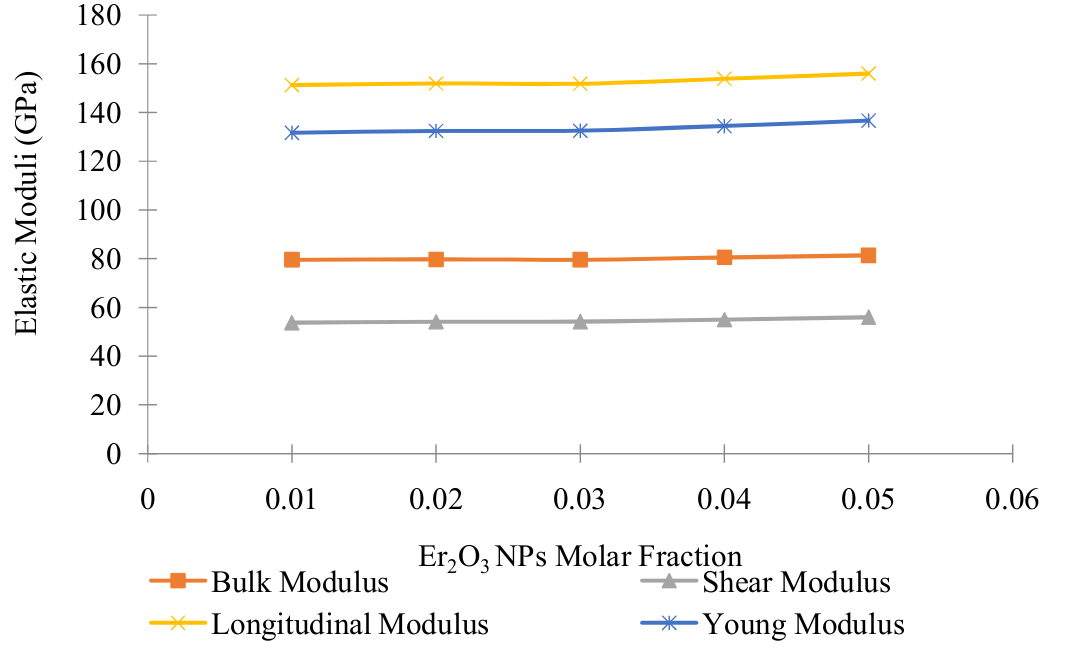
Elastic moduli of {[(TeO2)0.7 (B2O3)0.3]0.8 (SiO2)0.2}1-y (Er2O3 NPs)y glasses with y = 0.01, 0.02, 0.03, 0.04, 0.05 were studied in this work using the theoretical elastic models. The Makishima & Mackenzie, Rocherulle and bond compression models were employed for the study. In the Makishima and Mackenzie model, the packing density was calculated from the bulk glass molar weight and the bulk glass density whereas in Rocherulle model it is determined as the individual oxides. Young, shear and bulk moduli as well as the Poisson ratio were calculated for the glasses in the Makishima and Rocherulle models, while longitudinal, was calculated in addition to young, bulk and shear moduli using the bond compression model. Bond per unit volume number (nb), bulk modulus, bulk modulus ratio (Kbc/Ke), atomic ring size (?) and stretching force constant were also calculated and presented. The values of the Young, bulk and shear moduli obtained from Makishima model increased from 52.854 to 55.335 GPa, 35.754 to 39.862 GPa and 21.080 to 21.809 GPa respectively with Er2O3 NPs composition increase from 1% to 5%.. The Rocherulle model presented increasing values for Young, bulk and shear moduli as 56.910 to 58.432 GPa, 41.452 to 44.450 GPa and 22.385 to 22.809 GPa respectively with Er2O3 NPs composition increase from 1% to 5%. The bond compression model presented much higher values of the elastic moduli compared to the experimentally obtained values and showed an increasing trend as the Er2O3 NPs concentration increases. In the glass network, the atomic ring size value decreased from 0.5698 to 0.5091 nm indicating an increase in the close packing of atoms. Based on the elastic moduli values presented by all the models, Makishima and Mackenzie model presented a more reliable data and hence represents the best model for the studied glass system.
Published
How to Cite
Issue
Section
Copyright (c) 2022 Journal of the Nigerian Society of Physical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Similar Articles
- Oluwole Adigun, Lasisi Egibunu Umoru, Temidayo Nancy Iwatan, Effects of different electrolytes on the structure and yield ofgraphene oxide produced via electrochemical exfoliation , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 4, November 2023
- Ibrahim Adamu Mohammed, Majid Khan Majahar Ali, Sani Rabiu, Raja Aqib Shamim, Shahida Shahnawaz, Development and validation of hybrid drying kinetics models with finite element method integration for black paper in a v-groove solar dryer , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- Ammar A. Oglat, Abdallah Al Said, Naser M. Ahmed, Mohammed Dawood Salman, Zinc oxide nanoparticles and nanorods: advanced sunscreen ingredients for enhanced UV protection and radiation filtration , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- Sunday Olorunfunmi, Armand Bahini, Adenike Olatinwo, Theoretical Study on 10C Elastic Scattering Cross Sections Using Different Cluster Density Distributions and Different Potentials , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 2, May 2023
- Olumide Sunday Adesina, Bayesian Multilevel Models for Count Data , Journal of the Nigerian Society of Physical Sciences: Volume 3, Issue 3, August 2021
- Paavithashnee Ravi Kumar, Majid Khan Majahar Ali, Olayemi Joshua Ibidoja, Identifying heterogeneity for increasing the prediction accuracy of machine learning models , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 3, August 2024
- Namarta Singh, Jatinder saini, Effect of radial non-uniformity on mechanical response of functionally graded discs , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 1, February 2026
- Suraj Sharma, Ravinder Kumar, Analysis of love-type surface waves in an isotropic thermoelastic layer over a non-homogeneous elastic half-space with interface irregularity , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 2, May 2026
- C. A. Onate, I. B. Okon, E. S. Eyube, E. Omugbe, A. D. Ahmed, Computation of vibrational partition function: a comparative analysis between Poisson summation and classical limit , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 1, February 2026
- W. A. Yahya, A. A. Yahaya, A. A. Adewale, A. A. Sholagberu, N. K. Olasunkanmi, A DFT study of optoelectronic, elastic and thermo-electric properties of the double perovskites Rb2SeX6 (X=Br,Cl) , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 2, May 2023
You may also start an advanced similarity search for this article.






