Effect of Substitutional Point Defect of Gold (Au) in Indium (In) Site of Double Halide Perovskite (Cs2InSbCl6)
Keywords:
Perovskite, Photovoltaic solar cell, Optical, StructuralAbstract
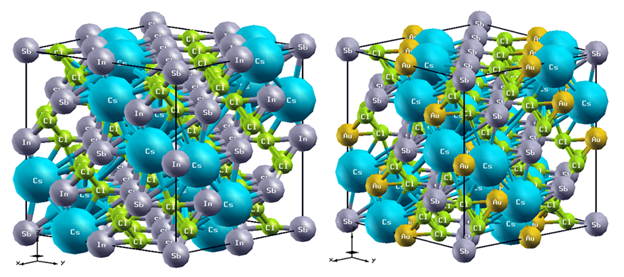
Lead (Pb) free (non-toxic) perovskite solar cells materials have attracted great interest in the commercialization of the photovoltaic devices. In this work, density functional theory (DFT) and linear response time-dependent within density functional theory (TDDFT) are used to simulate and investigate the effect of gold (Au) dopedPb-free double halide perovskite A2BB'X6(A = Cs; B = In, Au; B' = Sb; X = Cl) on the structural, electronic, and optical properties for perovskite solar cell application. On the structural properties, bond length and bulk modulus calculations show that the doped compound is more likely to resist deformation than the undoped compound. The calculated band structure for both materials (doped and undoped) reveals the presence of the Valence Band Maximum (VBM) and the Conduction Band Minimum (CBM) at around the same symmetry point which indicates a direct band gap nature (at Gamma point). The band gap value for the initial compound (Eg= 0.99 eV) agrees with published theoretical values. For the gold doped compound, the value of the band gap increased to a value of 1.25 eV. The result of the optical properties shows that the Au-doped material has higher absorption coefficient, lower reflectivity and higher optical conductivity when compared with the initial, as such demonstrates better properties as a candidate for solar cell applications and in other optoelectronic devices.
Published
How to Cite
Issue
Section
Copyright (c) 2021 Journal of the Nigerian Society of Physical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Similar Articles
- Essodossomondom Anate, N’Detigma Kata, Hodo-Abalo Samah, Amadou Seidou Maiga, Study of the passivation of defects in the perovskite cell: application to Sahelian climate conditions , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 2, May 2023
- Rafiu Adewale Busari, Ezekiel Omotoso, Lukman O. Animasahun, Saheed Adekunle Adewinbi, Emmanuel O. Adewumi, Comfort T. Famoroti, Bidini A. Taleatu, Adeniyi Y. Fasasi, Tuning the optical properties and some surface structure of Cd-O thin film electrodeposited by two-electrode: An effect of Cobalt incorporation , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 2, May 2023
- Muteeu A. Olopade, Anthony B. Adegboyega, Kayode I. Ogungbemi, Adeyinka D. Adewoyin, Investigation of the behaviour of tunable chalcogenide-Bismuth based perovskite BiTl (SxSe1-x)3(X = 0, 0.33, 0.67, 1): first principles calculations , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 1, February 2025
- W. A. Yahya, A. A. Yahaya, A. A. Adewale, A. A. Sholagberu, N. K. Olasunkanmi, A DFT study of optoelectronic, elastic and thermo-electric properties of the double perovskites Rb2SeX6 (X=Br,Cl) , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 2, May 2023
- I. T. Bello, Y. A. Odedunmoye, O. Adedokun, H. A. Shittu, A. O. Awodugba, Numerical Simulation of Sandwiched Perovskite-Based Solar Cell Using Solar Cell Capacitance Simulator (SCAPS-1D) , Journal of the Nigerian Society of Physical Sciences: Volume 1, Issue 2, May 2019
- D. Eli, M. Y. Onimisi, S. Garba, R. U. Ugbe, J. A. Owolabi, O. O. Ige, G. J. Ibeh, A. O. Muhammed, Simulation and Optimization of Lead-Based Perovskite Solar Cells with Cuprous Oxide as a P-type Inorganic Layer , Journal of the Nigerian Society of Physical Sciences: Volume 1, Issue 2, May 2019
- Odilon Joseph TOWANOU, Hagninou Elagnon Venance Donnou, Gabin Koto N’Gobi, Augustin Enonsi Leode, Basile Kounouh´ewa, Solar Energy Storage by Fuel Cell Technology at Abomey-Calavi (Benin) , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 2, May 2023
- Baba Alfa, Yakubu Adamu, Daniel Alberto Pena Perez, Optimal Power Point on the I-V Curve of a Photovoltaic Solar System (Modelling and Analysis) , Journal of the Nigerian Society of Physical Sciences: Volume 2, Issue 2, May 2020
- C. O. Lawani , G. J. Ibeha, Olumide Ige, D. Eli, J. O. Emmanuel, A. J. Ukwenya, P. O. Oyedare, Numerical Simulation of Copper Indium Gallium Diselenide Solar Cells Using One Dimensional SCAPS Software , Journal of the Nigerian Society of Physical Sciences: Volume 3, Issue 2, May 2021
- A. A. Faremi, S. S. Oluyamo, K. D. Adedayo, Y. A. Odusote, O. I. Olusola, Influence of Silicon Nanoparticle on the Electrical Properties of Heterostructured CdTe/CdS thin films based Photovoltaic Device , Journal of the Nigerian Society of Physical Sciences: Volume 3, Issue 3, August 2021
You may also start an advanced similarity search for this article.






