Corrosion Inhibition Potential of Thiosemicarbazide Derivatives on ALuminium: Insight from Molecular Modelling and QSARs Approaches
Keywords:
DFT, corrosion inhibitors, QSARs, ThiosemicarbazideAbstract
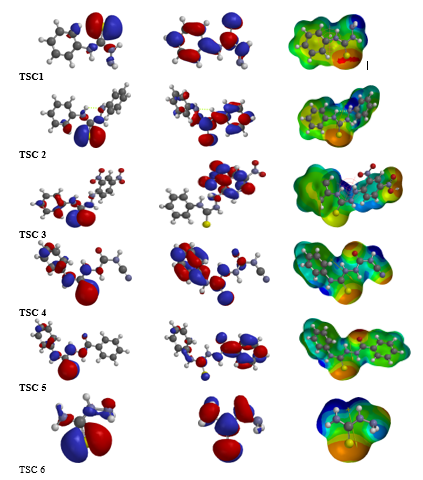
The potentials of six thiosemicarbazide derivatives towards corrosion inhibition were investigated theoretically using density functional theory (DFT) and quantitative structural-activity relationships (QSARs) methods. Their performance as corrosion inhibitors were evaluated using their calculated quantum chemical parameters such as molecular weight, softness, electronegativity, dipole moments, hardness, bandgap energy (\Delta E), highest occupied molecular orbital energy (EHOMO), and the lowest unoccupied molecular orbital energy (ELUMO). Regression analysis was carried out using the ordinary least square method to develop a model that establishes the relationship between chemical parameters and inhibition efficiencies that have been measured experimentally. According to the results, quantum chemical parameters confirm the inhibition potential of TSC5 to be greater than TSC2, while the predicted inhibition efficiencies of the studied thiosemicarbazide derivatives correspond to experimentally reported values with a root mean square error (%) of 1.116 and correlation coefficient of 0.998. The high correlation demonstrates and validates the quantum chemical approach’s reliability in studying corrosion inhibition on a metal surface. The validation of the developed model internally and externally demonstrates that it is robust and stable, with high predictability
Published
How to Cite
Issue
Section
Copyright (c) 2023 B. T. Ogunyemi, F. K. Ojo

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Similar Articles
- Saprizal Hadisaputra, Lalu Rudyat Telly Savalas, Corrosion Inhibition Properties of Lawsone Derivatives againts Mild Steel: A Theoretical Study , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 3, August 2023
- K. K. Adama, I. B. Onyeachu, The corrosion characteristics of SS316L stainless steel in a typical acid cleaning solution and its inhibition by 1-benzylimidazole: Weight loss, electrochemical and SEM characterizations , Journal of the Nigerian Society of Physical Sciences: Volume 4, Issue 2, May 2022
- Yazid Amri, Mohammed Abdelkader Belalem, Nedjimi Mohammed Said, Guerguer Louiza, Salah Tlili, Corrosion inhibition of carbon steel XC70 in 1M HCl solution using Balanite Aegyptiaca extracts as an eco-friendly inhibitor , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- C. B. Adindu, S. C. Nwanonenyi, C. B. C. Ikpa, Experimental and computational studies of the corrosion inhibitive effects of Zingiber officinale rhizomes on mild steel corrosion in acidic solutions , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 3, August 2023
- Titus O. Martins, Edwin A. Ofudje, Abimbola A. Ogundiran, Ojo A. Ikeoluwa, Osipitan A. Oluwatobi, Ezekiel F. Sodiya, Opeyemi Ojo, Cathodic Corrosion Inhibition of Steel by Musa Paradisiaca Leave Extract , Journal of the Nigerian Society of Physical Sciences: Volume 4, Issue 4, November 2022
- A. Shamsudeen, Shuaibu S., S.G. Abdu, M. S. Abubakar, Abdullahi lawal, First-principles calculations of Fluorine-doped Titanium dioxide: A prospective material for solar cells application , Journal of the Nigerian Society of Physical Sciences: Volume 1, Issue 4, November 2019
- F. L. Kherfane, A. BOUKRAA, B. Beladel, A. Douara, I. E. Tibermacine, A. Rabehi, M. Benghanem, Structural, electronic and optical properties of theFeAs(1-x)La(x) ternary alloys: a first principles calculations , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 1, February 2026
- W. A. Yahya, A. A. Yahaya, A. A. Adewale, A. A. Sholagberu, N. K. Olasunkanmi, A DFT study of optoelectronic, elastic and thermo-electric properties of the double perovskites Rb2SeX6 (X=Br,Cl) , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 2, May 2023
- T. M. J. Abdulkadhim, S. A. A. Alsaati, M. H. Shinen, Theoretical Investigation of Diameter Effects and Edge Configuration on the Optical Properties of Graphdiyne Nanotubes in the Presence of Electric Field , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 2, May 2023
- Segun Oladipo, Adesola A. Adeleke, Abosede A. Badeji, Katherine I. Babalola, Ayomide H. Labulo, Ibrahim Hassan, Sadiq T. Yussuf, Samuel O. Olalekan, Computational investigation and biological activity of selected Schiff bases , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 3, August 2024
You may also start an advanced similarity search for this article.