Modelling the transmission dynamics of Omicron variant of COVID-19 in densely populated city of Lagos in Nigeria
Keywords:
Epidemiological model, deterministic modelAbstract
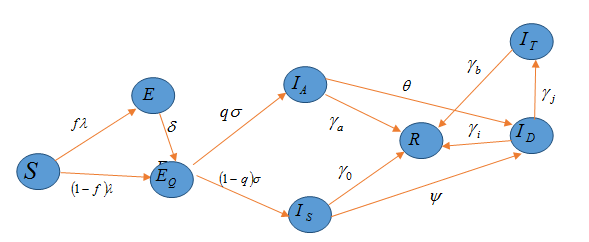
The kernel of the work in this article is the proposition of a model to examine the effect of control measures on the transmission dynamics of Omicron variant of coronavirus disease in the densely populated metropolis of Lagos. Data as relate to the pandemic was gathered as officially released by the Nigerian authority. We make use of this available data of the disease from 1st of December, 2021 to 20th of January, 2022 when omicron variant was first discovered in Nigeria. We computed the basic reproduction number, an epidemiological threshold useful for bringing the disease under check in the aforementioned geographical region of the country. Furthermore, a forecasting tool was derived, for making forecasts for the cumulative number of cases of infection as reported and the number of individuals where the Omicron variant of COVID-19 infection is active for the deadly disease. We carried out numerical simulations of the model using the available data so gathered to show the effects of non-pharmaceutical control measures such as adherence to common social distancing among individuals while in public space, regular use of face masks, personal hygiene using hand sanitizers and periodic washing of hands with soap and pharmaceutical control measures, case detecting via contact tracing occasioning clinical testing of exposed individuals, on the spread of Omicron variant of COVID-19 in the city. The results from the numerical simulations revealed that if detection rate for the infected people can be increased, with majority of the population adequately complying with the safety protocols strictly, then there will be a remarkable reduction in the number of people being afflicted by the scourge of the highly communicable disease in the city.
Published
How to Cite
Issue
Section
Copyright (c) 2023 Bolarinwa Bolaji, B. I. Omede, U. B. Odionyenma, P. B. Ojih, Abdullahi A. Ibrahim

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Similar Articles
- Bolarinwa Bolaji, Abdullahi Ibrahim, Favour Ani, Benjamin Omede, Godwin Acheneje, A model for the control of transmission dynamics of human monkeypox disease in Sub-Saharan Africa , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 2, May 2024
- Obiora Cornelius Collins, Mathematical model analysis for maize yield under co-infection of maize streak virus and maize stripe virus diseases with control measures , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 2, May 2026
- E. A. Nwaibeh, M. K. M. Ali, M. O. Adewole, The dynamics of hybrid-immune and immunodeficient susceptible individuals and the three stages of COVID-19 vaccination , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 3, August 2024
- Saheed Ajao, Isaac Olopade, Titilayo Akinwumi, Sunday Adewale, Adelani Adesanya, Understanding the Transmission Dynamics and Control of HIV Infection: A Mathematical Model Approach , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 2, May 2023
- Ogechi Regina Amanso, Jeconia Okelo Abonyo, Phineas Roy Kiogora, Obiora Cornelius Collins, A novel mathematical model for transmission dynamics of HPV and cervical cancer progression with cancer-reliant awareness , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 2, May 2026
- Samson Olaniyi, Furaha M. Chuma, Sulaimon F. Abimbade, Asymptotic stability analysis of a fractional epidemic model for Ebola virus disease in Caputo sense , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 1, February 2025
- Kazeem A. Tijani, Chinwendu. E. Madubueze, Isaac O. Onwubuya, Nkiruka Maria-Assumpta Akabuike, John Olajide Akanni, Mathematical modelling of the dynamical system of military population, focusing on the impact of welfare , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- Gurpreet Singh Tuteja, Tapshi Lal, A study of growth of COVID-19 with super-spreaders using the modified SIR model including iceberg phenomenon , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 2, May 2024
- James Andrawus, Kayode Isaac Omotoso, Agada Apeh Andrew, Felix Yakubu Eguda, Sunday Babuba, Kabiru Garba Ibrahim, Mathematical model analysis on the significance of surveillance and awareness on the transmission dynamics of diphtheria , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- Tunde Tajudeen Yusuf, Afeez Abidemi, Ayodeji Sunday Afolabi, Emmanuel Jesuyon Dansu, Optimal Control of the Coronavirus Pandemic with Impacts of Implemented Control Measures , Journal of the Nigerian Society of Physical Sciences: Volume 4, Issue 1, February 2022
You may also start an advanced similarity search for this article.
Most read articles by the same author(s)
- Bolarinwa Bolaji, Abdullahi Ibrahim, Favour Ani, Benjamin Omede, Godwin Acheneje, A model for the control of transmission dynamics of human monkeypox disease in Sub-Saharan Africa , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 2, May 2024






