Optimization by Box Behnken Design for Eosin Yellow Dye Removal from Aqueous Medium using Date Palm Seeds-Porous Carbon@TiO2 Blend
Keywords:
Porous carbon, TiO2, Adsorption, Eosin yellow dye, Box Behnken designAbstract
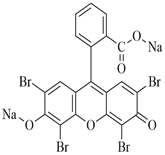
Biological stains are potentially harmful compounds present in the environment, in which Eosin yellow dye (EYD) is one of the most commonly applied stains. In this research, date palm seeds-porous carbon (DPSC) and its TiO2 blend (TiO2-DPSC) were prepared and their efficiency on the removal of EYD from an aqueous medium was investigated. Characterization by SEM, EDX, FTIR and BET surface area was performed on the materials. The BET surface area (542.63 m2/g) and pore diameter (2.02 nm) of TiO2-DPSC were found to be higher than that of DPSC (332.74 m2/g and 1.85 nm) indicating that TiO2-DPSC is mesoporous while DPSC is microporous. The major and interactive impacts of the adsorption parameters: initial EYD concentration, pH, adsorbent dose, and time of contact were examined by Box Behnken design in response surface methodology. The high R2 values 0.9658 and 0.9597 for DPSC and TiO2-DPSC agreed with the adjusted R2 values suggesting the quadratic model sufficiently interprets the adsorption data. The optimum removal efficiency of EYD onto DPSC and TiO2-DPSC was 34.63 mg/g and 55.34 mg/g which are in agreement with the predicted removal of 34.75 mg/g and 50.11 mg/g respectively at the center point values of Co=300 mg/L, pH 2, 362.5 min and 0.1 g adsorbent dose. The results also showed the acceptability of the Box Behnken design in response surface methodology for the optimization of EYD removal from aqueous media using DPSC and TiO2-DPSC blends. Hence, better EYD removal reported in TiO2-DPSC compared to DPSC was due to its improved adsorptive features.
Published
How to Cite
Issue
Section
Copyright (c) 2022 Journal of the Nigerian Society of Physical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Similar Articles
- K. O. Sodeinde, S. O. Olusanya, D. U. Momodu, V. F. Enogheghase, O. S. Lawal, Waste glass: An excellent adsorbent for crystal violet dye, Pb2+ and Cd2+ heavy metals ions decontamination from wastewater , Journal of the Nigerian Society of Physical Sciences: Volume 3, Issue 4, November 2021
- C. J. Ajaelu, O. Oyedele, A. A. Ikotun, E. O. Faboro, Safranin O dye removal using Senna fistula activated biomass: Kinetic, equilibrium and thermodynamic studies , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 1, February 2023
- Abdullahi Moyosore, Haslina Ahmad, Muhammad Alif Muhammad Latif, Mostafa Yousefzadeh Borzehandani, Mohd Basyaruddin AbdulRahman, Emilia Abdelmalek, Carbon (IV) oxide adsorption efficiency of functionalized HKUST-1, IRMF-1, and UiO-66 metal organic frameworks , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 1, February 2024
- Emma Panzi Mukhokosi, Stephen Tenywa, Nandipha L. Bothab, Shohreh Azizi, Mathapelo Pearl Seopela, Malik Maaza, Green synthesis of CuO nanoparticles from Cucurbita maxima leaf extract; a platinum free counter electrode for dye sensitized solar cells , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 1, February 2025
- S. A. Adesokan, A. A. Giwa, I. A. Bello, Removal of Trimethoprim from Water using Carbonized Wood Waste as Adsorbents , Journal of the Nigerian Society of Physical Sciences: Volume 3, Issue 4, November 2021
- M. Siddalinga Prasad, An exploration of thermal characteristics of a permeable inclined moving inverted exponential fully wet magnetized stretching/shrinking fin , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- A. H. Labulo, A. D. Terna, O. F. Oladayo, H. Ibrahim, N. S. Tanko, R. A. Ashonibare, J. D. Opeyemi, Z. Tywabi-Ngeva, Photocatalytic and antibacterial activities of green-mediated Khaya senegalensis-silver nanoparticles and oxidized carbon nanotubes , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 3, August 2023
- Yazid Amri, Mohammed Abdelkader Belalem, Nedjimi Mohammed Said, Guerguer Louiza, Salah Tlili, Corrosion inhibition of carbon steel XC70 in 1M HCl solution using Balanite Aegyptiaca extracts as an eco-friendly inhibitor , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- M. Ramanuja, J. Kavitha, A. Sudhakar, N. Radhika, Study of MHD SWCNT-Blood Nanofluid Flow in Presence of Viscous Dissipation and Radiation Effects through Porous Medium , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 1, February 2023
- Saprizal Hadisaputra, Lalu Rudyat Telly Savalas, Corrosion Inhibition Properties of Lawsone Derivatives againts Mild Steel: A Theoretical Study , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 3, August 2023
You may also start an advanced similarity search for this article.






