Mathematical Model of In-host Dynamics of Snakebite Envenoming
Keywords:
Snakebite, in-host model, venom, antivenom, stability analysisAbstract
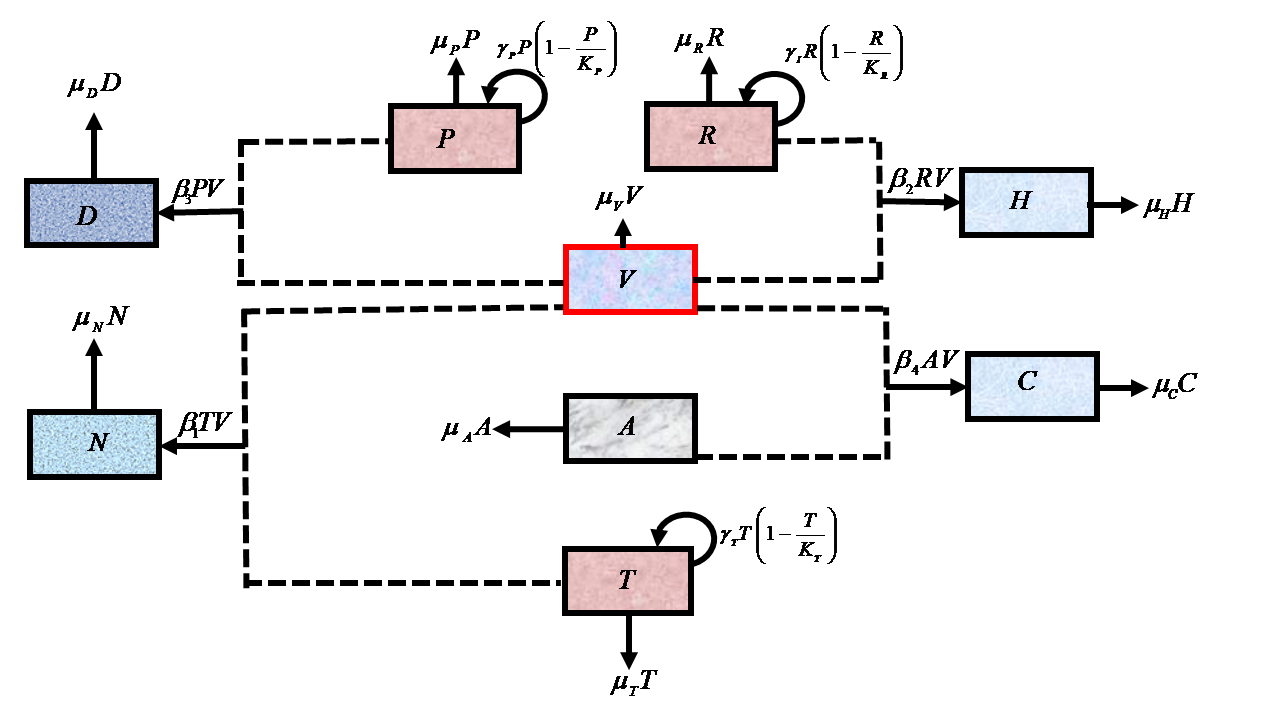
In this paper, we develop an in-host mathematical model of snakebite envenoming that includes tissue, red blood and platelet cells of humans as specific targets of different kinds of toxins in the snake venom. The model is use to study some harmful effects of cytotoxic and hemotoxic snake venom on their target cells under the influence of snake antivenom. The model has two equilibrium points, namely, trivial and venom free. It has been shown that both the equilibrium points are globally asymptotically stable and numerical simulations illustrate the global asymptotic stability of the venom free equilibrium point. Furthermore, simulations reveal the importance of administering antivenom to avert the possible damage from venom toxins on the target cells. It is also shown through simulation that administering the required dose of antivenom can lead to the elimination of venom toxins within one week. Therefore, we recommend the administration of an adequate dose of antivenom therapy as it helps in deactivating venom toxins faster and consequently enhances the recovery time.
Published
How to Cite
Issue
Section
Copyright (c) 2022 Journal of the Nigerian Society of Physical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Similar Articles
- Ogechi Regina Amanso, Jeconia Okelo Abonyo, Phineas Roy Kiogora, Obiora Cornelius Collins, A novel mathematical model for transmission dynamics of HPV and cervical cancer progression with cancer-reliant awareness , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 2, May 2026 (In Progress)
- Kazeem A. Tijani, Chinwendu. E. Madubueze, Isaac O. Onwubuya, Nkiruka Maria-Assumpta Akabuike, John Olajide Akanni, Mathematical modelling of the dynamical system of military population, focusing on the impact of welfare , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- J. Andrawus, J. Y. Musa, S. Babuba, A. Yusuf, S. Qureshi, U. T. Mustapha, A. Oghenefejiro, I. S. Mamba, Modeling the dynamics of pertussis to assess the influence of timely awareness with optimal control analysis , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- Obiora Cornelius Collins, Mathematical model analysis for maize yield under co-infection of maize streak virus and maize stripe virus diseases with control measures , Journal of the Nigerian Society of Physical Sciences: Volume 8, Issue 2, May 2026 (In Progress)
- Samson Olaniyi, Furaha M. Chuma, Sulaimon F. Abimbade, Asymptotic stability analysis of a fractional epidemic model for Ebola virus disease in Caputo sense , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 1, February 2025
- James Andrawus, Kayode Isaac Omotoso, Agada Apeh Andrew, Felix Yakubu Eguda, Sunday Babuba, Kabiru Garba Ibrahim, Mathematical model analysis on the significance of surveillance and awareness on the transmission dynamics of diphtheria , Journal of the Nigerian Society of Physical Sciences: Volume 7, Issue 4, November 2025
- L. Adamu, N. Hussaini, An Epidemic Model of Zoonotic Visceral Leishmaniasis with Time Delay , Journal of the Nigerian Society of Physical Sciences: Volume 1, Issue 1, February 2019
- Peter Urane Achimugwu, Mathew Ngugi Kinyanjui, David Mumo Malonza, Analysis of a fractional order climate model due to excessive emission and accumulation of carbon dioxide in the atmosphere , Journal of the Nigerian Society of Physical Sciences: Volume 5, Issue 4, November 2023
- Solomon A. Ayuba, I. Akeyede, A. S. Olagunju, Stability and Sensitivity Analysis of Dengue-Malaria Co-Infection Model in Endemic Stage , Journal of the Nigerian Society of Physical Sciences: Volume 3, Issue 2, May 2021
- A. O. Sangotola, S. B. Adeyemo, O. A. Nuga, A. E. Adeniji, A. J. Adigun, A tuberculosis model with three infected classes , Journal of the Nigerian Society of Physical Sciences: Volume 6, Issue 1, February 2024
You may also start an advanced similarity search for this article.
Most read articles by the same author(s)
- L. Adamu, N. Hussaini, An Epidemic Model of Zoonotic Visceral Leishmaniasis with Time Delay , Journal of the Nigerian Society of Physical Sciences: Volume 1, Issue 1, February 2019






